 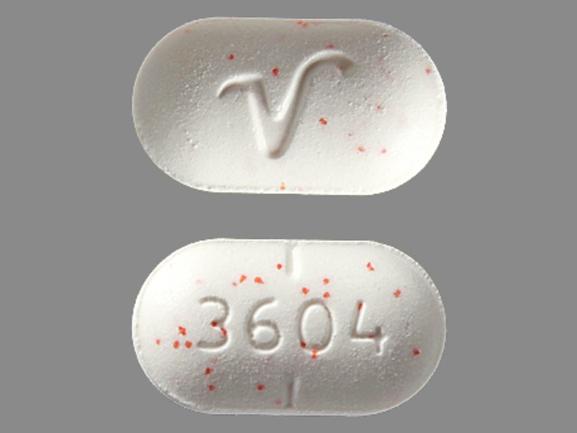 
The FDA encourages drugmakers to include a letter or a number in a drug’s imprint code because those digits can help health care workers identify a pill more easily than a symbol or logo alone, the agency says. Once a drugmaker submits a pill’s imprint code to the FDA, the agency stores it in a database that health care professionals can access. It might include words, the drugmaker’s name, or other details. This code - combined with the pill’s size, color, and shape - helps you tell it apart from other medications.Ī pill’s imprint code can be made up of any single letter or number, or any combination of letters, numbers, marks, or symbols. The FDA requires most OTC and prescription drug tablets and capsules to have an imprint. The numbers, letters, or other text that you see on a pill are its “imprint code,” a feature that helps you identify it. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
